Rheology of liquids in the context of powder mixing
In this blog post, we will examine a key aspect of powder moistening: the influence of the rheological properties of different liquids on the mixing result. Rheology largely determines how a liquid flows, how it behaves under shear stress, and how well it can wet powder particles. These relationships have a direct impact on the appearance of the wetted powder – on its flowability, the homogeneity of the mixture, its tendency to agglomerate, and the preservation of the original particle shape and size.
In order to wet powder homogeneously, all particles must receive a comparable layer of liquid that corresponds to their specific surface area. The aim of the mixing process is therefore to distribute both small and large quantities of liquid evenly over the enormous total contact surface of the powder. After the mixing process, there should be no areas of over-moistening or dryness. The achievable product quality is largely determined by the interaction of defined droplet size, precise dosing strategy and adapted mixing energy. In this context, it is advantageous to have a good understanding of the chemical and physical properties of the wetting liquid.
Viscosity and temperature dependence
Dynamic viscosity η describes a liquid's resistance to shear or deformation and is a key parameter for mixing behaviour. For many low to medium viscosity liquids, the temperature dependence of viscosity can be approximated using an Arrhenius approach:
η(T) = η0 * exp ( Eη / (R * T) )
with
η … dynamic viscosity,
η0 … material constant (viscosity at reference temperature),
Eη … activation energy of the flow process,
R … universal gas constant,
T … absolute temperature in Kelvin.
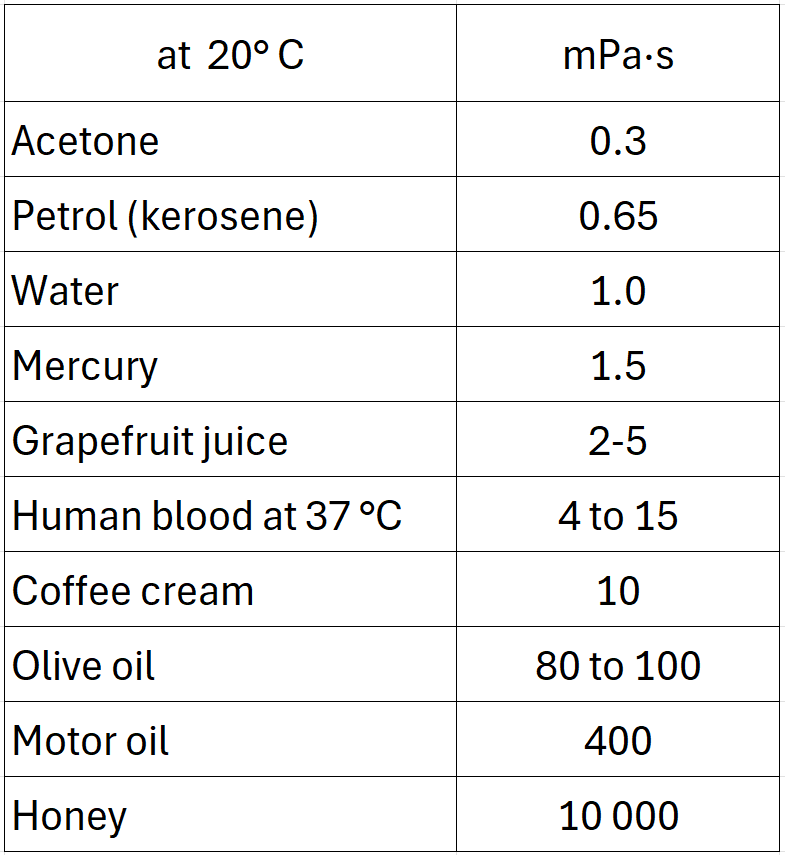
As the temperature rises, the viscosity of most organic liquids decreases significantly, so that highly viscous components such as lecithins, molasses, honey, oleoresins or certain vegetable oils can be pumped, finely dosed and distributed in the mixing chamber more easily by moderate heating. In many cases, the changes in viscosity are reversible. However, in the case of complex liquids (e.g. emulsions, concentrated sugar solutions or structurally viscous systems), thermally or mechanically induced structural changes can lead to hysteresis effects, meaning that the flow behaviour is not identical during heating and cooling.
Dynamic viscosity η describes a liquid's resistance to shear or deformation and is a key parameter for mixing behaviour. For many low to medium viscosity liquids, the temperature dependence of viscosity can be approximated using an Arrhenius approach:
η(T) = η0 * exp ( Eη / (R * T) )
with
η … dynamic viscosity,
η0 … material constant (viscosity at reference temperature),
Eη … activation energy of the flow process,
R … universal gas constant,
T … absolute temperature in Kelvin.
As the temperature rises, the viscosity of most organic liquids decreases significantly, so that highly viscous components such as lecithins, molasses, honey, oleoresins or certain vegetable oils can be pumped, finely dosed and distributed in the mixing chamber more easily by moderate heating. In many cases, the changes in viscosity are reversible. However, in the case of complex liquids (e.g. emulsions, concentrated sugar solutions or structurally viscous systems), thermally or mechanically induced structural changes can lead to hysteresis effects, meaning that the flow behaviour is not identical during heating and cooling.
Influence of shear stress and shear rate
Liquids of different genera. Mostly the viscosity changes are reversible. But often with considerable hysteresis.
The interaction between shear stress τ and shear rate γ˙ is crucial for understanding behaviour in the mixer. In the simplest case, apparent viscosity can be defined as
η = τ / γ˙
where
η … apparent viscosity
τ … shear stress,
γ˙ … shear rate.
Liquids of different genera. Mostly the viscosity changes are reversible. But often with considerable hysteresis.
The interaction between shear stress τ and shear rate γ˙ is crucial for understanding behaviour in the mixer. In the simplest case, apparent viscosity can be defined as
η = τ / γ˙
where
η … apparent viscosity
τ … shear stress,
γ˙ … shear rate.
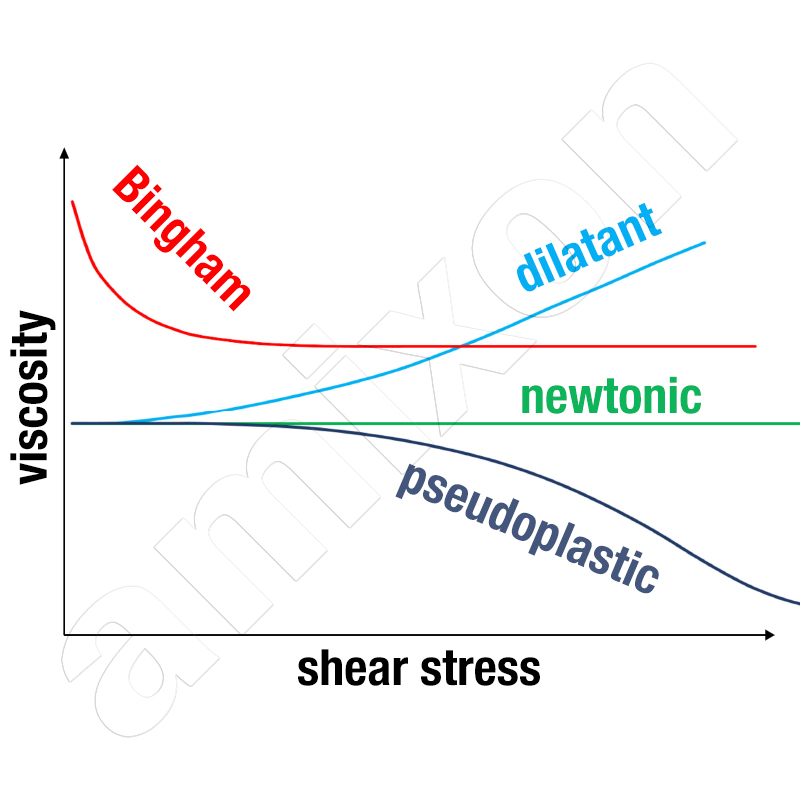
Newtonian fluids
In Newtonian fluids, viscosity is independent of shear rate:
τ = η * γ˙, η = constant
Typical examples are water, many cooking oils and highly diluted solutions. Their flow behaviour is linear and easily predictable. Surface tension can be reliably characterised. Viscoelastic effects do not occur. The viscosity only changes with temperature.
This makes Newtonian fluids particularly easy to control in mixing processes. They enable reproducible and uniform wetting – provided that the dosage and type of liquid addition are correctly designed.
In Newtonian fluids, viscosity is independent of shear rate:
τ = η * γ˙, η = constant
Typical examples are water, many cooking oils and highly diluted solutions. Their flow behaviour is linear and easily predictable. Surface tension can be reliably characterised. Viscoelastic effects do not occur. The viscosity only changes with temperature.
This makes Newtonian fluids particularly easy to control in mixing processes. They enable reproducible and uniform wetting – provided that the dosage and type of liquid addition are correctly designed.
Shear-thinning or pseudoplastic liquids
Shear-thinning or pseudoplastic liquids exhibit a decrease in apparent viscosity as the shear rate increases. Their behaviour can often be described by the Ostwald-de Waele or power law model. These liquids are generally very easy to spray. They can be sprayed using single- or multi-component atomising nozzles.
τ = K * γ˙n ; n < 1
with
γ˙ … shear rate
K … consistency index,
n … flow index (degree of shear thinning).
Examples include tomato paste, many gelling agents, polysaccharide solutions and numerous emulsions. Under the high local shear conditions in the mixing chamber, these liquids become more fluid, which facilitates their distribution on the powder surface. At the same time, they remain sufficiently viscous at rest to reduce settling or segregation.
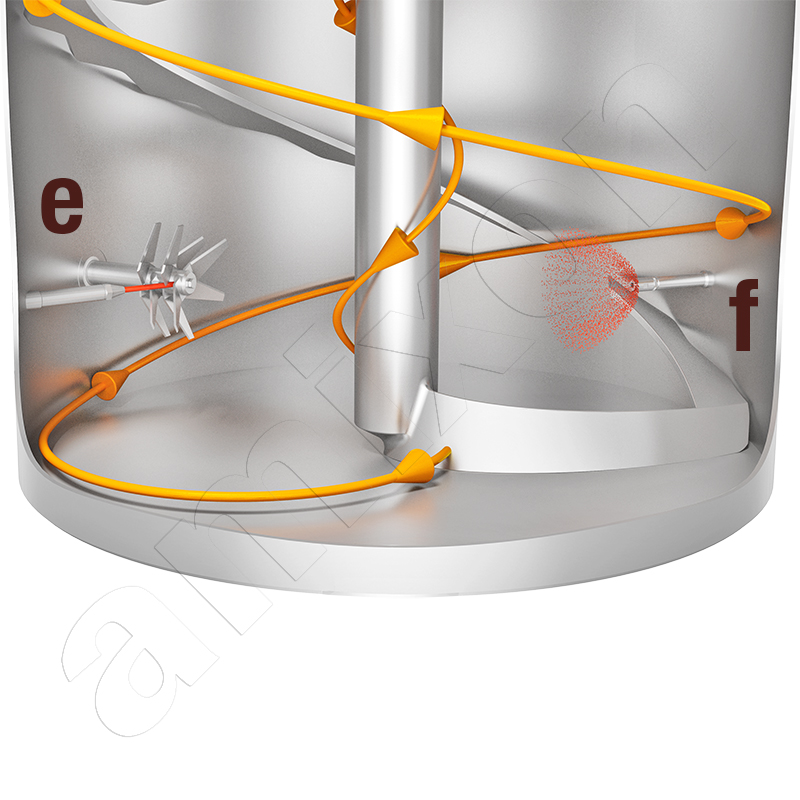
The amixon® cone mixer shown on the left mixes continuously. It stands on load cells. Several powders and a liquid component flow into the cone mixer in gravimetric doses. The discharge fitting is at the bottom. It continuously discharges the mixed goods so that the filling level remains constant. In this way, the powders are mixed very gently and moistened evenly. The AMK 600 continuous mixer is designed for 12 to 15 m³/h.
Shear-thinning or pseudoplastic liquids exhibit a decrease in apparent viscosity as the shear rate increases. Their behaviour can often be described by the Ostwald-de Waele or power law model. These liquids are generally very easy to spray. They can be sprayed using single- or multi-component atomising nozzles.
τ = K * γ˙n ; n < 1
with
γ˙ … shear rate
K … consistency index,
n … flow index (degree of shear thinning).
Examples include tomato paste, many gelling agents, polysaccharide solutions and numerous emulsions. Under the high local shear conditions in the mixing chamber, these liquids become more fluid, which facilitates their distribution on the powder surface. At the same time, they remain sufficiently viscous at rest to reduce settling or segregation.
The amixon® cone mixer shown on the left mixes continuously. It stands on load cells. Several powders and a liquid component flow into the cone mixer in gravimetric doses. The discharge fitting is at the bottom. It continuously discharges the mixed goods so that the filling level remains constant. In this way, the powders are mixed very gently and moistened evenly. The AMK 600 continuous mixer is designed for 12 to 15 m³/h.
Shear-thickening (dilatant) liquids
In shear-thickening (dilatant) liquids, the apparent viscosity increases with increasing shear rate. They are therefore sensitive to rapid movements and high local shear forces. Such media should be handled as slowly as possible and with minimal shear stress.
τ = K * γ˙n ; n > 1
Shear-thickening behaviour is typical for starch suspensions and highly concentrated dispersions. If these materials are conveyed quickly, the viscosity can increase sharply. In practice, good mixing results are usually achieved when the shear-thickening liquid is introduced into a zone of intense powder turbulence in a relaxed, low-viscosity state. In the powder mixer, it is finely distributed in this way before the dilatant viscosity increases.
Ring layer mixers can also be used for the continuous distribution of shear-thickening liquids. The mixing chamber consists of a horizontally arranged pipe. A fast-rotating mixing tool rotates inside it. The high rotation frequency creates a stable ring layer of compacted powder particles. Strong turbulence and intense shear forces are at work in this zone. Ring layer mixers can not only wet powders homogeneously. With the liquid dosage set correctly, they can also agglomerate the product evenly.
In shear-thickening (dilatant) liquids, the apparent viscosity increases with increasing shear rate. They are therefore sensitive to rapid movements and high local shear forces. Such media should be handled as slowly as possible and with minimal shear stress.
τ = K * γ˙n ; n > 1
Shear-thickening behaviour is typical for starch suspensions and highly concentrated dispersions. If these materials are conveyed quickly, the viscosity can increase sharply. In practice, good mixing results are usually achieved when the shear-thickening liquid is introduced into a zone of intense powder turbulence in a relaxed, low-viscosity state. In the powder mixer, it is finely distributed in this way before the dilatant viscosity increases.
Ring layer mixers can also be used for the continuous distribution of shear-thickening liquids. The mixing chamber consists of a horizontally arranged pipe. A fast-rotating mixing tool rotates inside it. The high rotation frequency creates a stable ring layer of compacted powder particles. Strong turbulence and intense shear forces are at work in this zone. Ring layer mixers can not only wet powders homogeneously. With the liquid dosage set correctly, they can also agglomerate the product evenly.
Practical process development at the amixon® Technical Center
The amixon® team cordially invites you to test your powdered products at the Technical Center. There, a wide variety of powders are modified and enhanced by moistening on an almost daily basis.
The addition of liquid must be optimally adapted to the properties of the powder and liquid. In addition, the temperature, concentration, and formulation must be adjusted. The defined sequence of material additions is equally crucial. The mixing intensity must also be correct without damaging the product. Sometimes it is advisable to distribute the liquids gradually in the powder. Multistep mixing processes are particularly effective, in which finer powder particles coat slightly over-moistened particles. This binds dust-fine fractions. This improves flowability, reduces dust pollution, and stabilizes the downstream packaging technology.
amixon® supports you in finding the optimal process control – precisely, quickly, and practically.
The amixon® team cordially invites you to test your powdered products at the Technical Center. There, a wide variety of powders are modified and enhanced by moistening on an almost daily basis.
The addition of liquid must be optimally adapted to the properties of the powder and liquid. In addition, the temperature, concentration, and formulation must be adjusted. The defined sequence of material additions is equally crucial. The mixing intensity must also be correct without damaging the product. Sometimes it is advisable to distribute the liquids gradually in the powder. Multistep mixing processes are particularly effective, in which finer powder particles coat slightly over-moistened particles. This binds dust-fine fractions. This improves flowability, reduces dust pollution, and stabilizes the downstream packaging technology.
amixon® supports you in finding the optimal process control – precisely, quickly, and practically.
Customer quote: ‘Our visit to amixon® was a great source of information....’
...our product and process development team gained valuable insights. That was a real surprise! We hadn't expected that before we set off on our trip."
At the amixon® technical centre, we not only mix and moisten your powders to the highest standard. amixon® equipment can also agglomerate, synthesise and vacuum dry. Always using state-of-the-art technology! There are more than 30 test machines to choose from. From 1 litre to 3 m³.
...our product and process development team gained valuable insights. That was a real surprise! We hadn't expected that before we set off on our trip."
At the amixon® technical centre, we not only mix and moisten your powders to the highest standard. amixon® equipment can also agglomerate, synthesise and vacuum dry. Always using state-of-the-art technology! There are more than 30 test machines to choose from. From 1 litre to 3 m³.
© Copyright by amixon GmbH