Hydrogen bonds
Hydrogen bonds are specific intermolecular interactions between a hydrogen atom and a highly electronegative atom, for example oxygen, nitrogen, or fluorine. The hydrogen atom is covalently bonded to an electronegative atom and additionally interacts with a lone electron pair of a neighboring molecule.
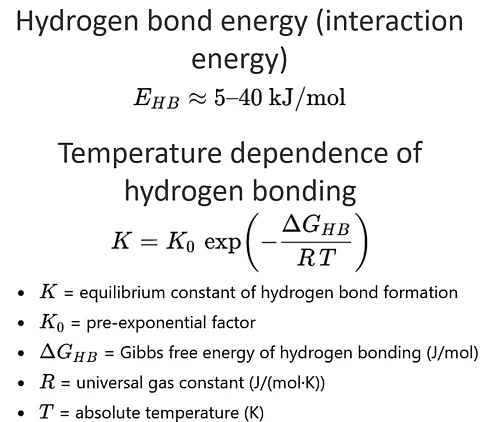
Hydrogen bond energy (typical range)
E_HB ≈ 5 – 40 kJ/mol
- E_HB is the hydrogen bond energy
- typical values depend on the donor-acceptor pair
Temperature dependence of hydrogen bonding (Arrhenius equilibrium relation)
K = K_0 * exp ( - DeltaG_HB / (R * T) )
- K is the equilibrium constant of hydrogen bond formation
- K_0 is a pre-exponential factor
- DeltaG_HB is the Gibbs free energy of hydrogen bonding (J/mol)
- R is the universal gas constant (J/(mol·K))
- T is the temperature in (K)
Hydrogen bonds are weaker than covalent bonds but significantly stronger than pure van der Waals forces. They lead to a directional attraction between molecules and influence their spatial arrangement. As a result, they shape many physical properties of substances.
In process engineering, hydrogen bonds play an important role in liquids, solutions, and polymers. They influence viscosity, gel formation, solubility, and swelling behavior. In particular, water, alcohols, starch, cellulose derivatives, and many thickeners exhibit pronounced hydrogen bonding.
Hydrogen bonds are temperature-dependent. With increasing temperature, they are partially disrupted. This explains decreases in viscosity, structural changes, and phase transitions upon heating. Mechanical shear can also temporarily disrupt hydrogen bonds.
During mixing, tempering, and drying, hydrogen bonds therefore significantly influence process behavior. They determine how substances interact with each other and how stable the formed material structures are.

