Enthalpy of vaporization
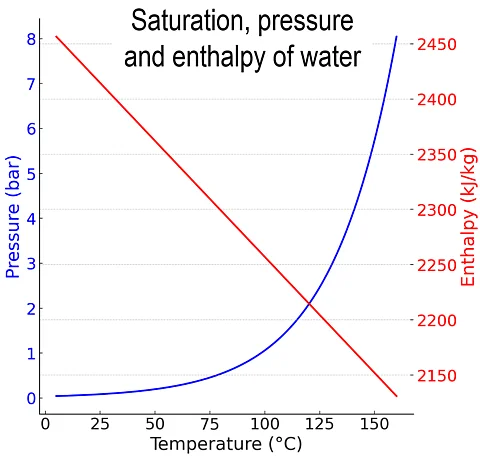
The enthalpy of vaporization describes the specific energy required to evaporate a liquid at constant temperature and constant pressure. It describes the phase transition from the liquid to the gaseous phase. The enthalpy of vaporization depends on the substance and on temperature.
In process engineering, the enthalpy of vaporization is a key parameter for drying processes, evaporators, distillation, and vacuum drying. It determines the energy demand of a process and influences the design of heating surfaces and energy supply.
The heat requirement for evaporation is described by the following equation:
Q_dot =m_dot * Delta_h_vap
- Q_dot = heat flow or heat requirement (W)
- m_dot = mass flow rate of the evaporated liquid (kg/s)
- Delta_h_vap = enthalpy of vaporization (J/kg))
For water at 100 °C, the enthalpy of vaporization is about 2257 kJ/kg and decreases with increasing temperature. Under vacuum, the enthalpy of vaporization remains nearly constant while the boiling point decreases.
In dryers and vacuum contact dryers, the enthalpy of vaporization is often the dominant share of energy. It determines the minimum theoretical energy required to dehumidify a product.

