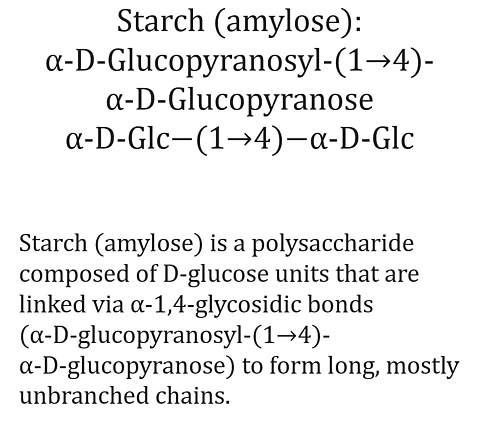
alpha-glycosidic bond
An α-glycosidic bond is a glycosidic bond in which the anomeric centre of the sugar involved is in the α-configuration. In the Haworth projection, this means that the hydroxyl group on the anomeric carbon atom (usually C-1) points downwards in D-sugars. Accordingly, the oxygen atom of the glycosidic bridge also points ‘downwards’, in the trans configuration to the CH₂₂OH group. Typical examples include the α-1,4-glycosidic bond in amylose and amylopectin, or the α-1,6-branches in starch and glycogen.
An α-glycosidic bond is a glycosidic bond in which the anomeric centre of the sugar involved is in the α-configuration (e.g. α-D-glucopyranosyl-(1→4)-α-D-glucopyranose). In the Haworth projection, this means that the hydroxyl group on the anomeric carbon atom (usually C-1) points downwards in D-sugars. Accordingly, the bonding oxygen atom of the glycosidic bridge also points ‘downwards’, trans to the CH₂OH group. Typical examples include the α-1,4-glycosidic bond in amylose and amylopectin, or the α-1,6-branches in starch and glycogen.

