Powder mixture
A powder mixture consists of at least two different solids. The solids are present in powder form, i.e. dispersed. The individual particles are physically separated from one another and no chemical reaction takes place. Powder mixtures are of central importance in almost all industries, for example in the chemical, food, pharmaceutical and materials industries. One quality parameter is the homogeneity of the distribution of the substances, i.e. the mixing quality.
A powder mixture is considered ideally mixed when a statistical random distribution of all particles is achieved. In this state, the probability of finding a specific particle at any position in the mixture is the same everywhere; this distribution is referred to as an ideal mixture.
The ideal random distribution can be described mathematically, provided the particle size distribution of all components is known and the sample size has been correctly defined.
The statistical variation in the concentration of a component in a sample is given by the variance of a Bernoulli-distributed random variable:
σ² = (p · (1 − p)) / n
- p is the mass or volume fraction of the component in question
- n is the number of particles in the sample
As the sample size n increases, the statistical dispersion σ decreases; the quality of mixing is therefore always scale-dependent. A real powder mixture cannot be more homogeneous than its ideal random distribution; this represents the physical upper limit of miscibility.
In practice, the quality of mixing is often described using the coefficient of variation CV:
CV = σ / μ
- σ is the standard deviation of the concentration
- μ is the mean concentration value
A small coefficient of variation indicates a high quality of mixing. As a relative parameter, the CV is dimensionless and therefore independent of physical units. In many mixtures, values below approximately 5 per cent are considered very good, although the exact thresholds may vary depending on the particle structure. In the literature, the following ranges are typically cited for component ratios of 1:100 or 1:1000:
- CV > 10% → poorly mixed
- CV ≈ 5% → well mixed
- CV < 2% → very well mixed; conditions are particularly good. This is very rarely the case in practice.
In special cases, component ratios as high as 1:100,000 are even mixed to test the performance of powder mixers. The actual mixing quality achievable depends heavily on the material properties of the components involved and on the mixing principle employed. Low-shear, three-dimensional mixing mechanisms with controlled relative particle movements facilitate the achievement of ideal distribution.
Factors influencing mixability
Certain properties of the components promote particularly homogeneous mixing and reduce the tendency for segregation:
- A uniform particle size reduces segregation effects; large differences favour percolation and segregation.
- A particle shape that is as round as possible improves flow properties and reduces mechanical entanglement.
- Similar bulk densities prevent gravitational segregation; pronounced differences in density have a separating effect.
- Comparable flow properties support uniform relative movements; significant differences lead to flow decoupling.
- A narrow particle size distribution increases statistical uniformity; broad distributions hinder homogenisation.
- Uniform wetting can reduce dust formation and electrostatic effects and stabilise the mixture.
- Similar surface energies and interactions between components are advantageous; strongly differing affinities promote segregation.
- Agglomerates behave like independent coarse particles; their disagglomeration down to the primary particles is crucial if a fine dispersion is desired.
- Other influencing factors include temperature, moisture content, mixing time, fill level and the type of flow through the mixer.
- Electrostatic charging leads to the separation of fine dust and impairs the mixing quality.
Minimum sample size for the homogeneity test
A sufficient sample size is required for the evaluation of mixing quality. The minimum number n_min of particles in a sample can be derived from the desired maximum concentration variation:
n_min = (1 − p) / (p · CV_max²)
- p is the mass or volume fraction of the component under consideration
- CV_max is the maximum permissible coefficient of variation
Direct particle counting is rarely feasible in practice, which is why the corresponding minimum sample mass m_p is required. For this, the average particle mass is required, which, assuming spherical particles, is calculated from the density and average particle diameter:
m_p = ρ · (π · d³) / 6
- m_p is the minimum sample mass
- ρ is the particle density
- π is the mathematical constant
- d is the mean particle diameter
Example:
Even at a mass fraction of less than 1 per cent, the literature refers to a trace component. If the coefficient of variation (CV) for the mixing quality of this component is to be a maximum of 5 per cent, the required minimum particle count n_min in the sample is given by the equation:
- Proportion p=0.01
- Desired CVmax=0.05
n_min= 1/(0.01⋅0.052) = 40,000
Mixing quality can be expressed as a dimensionless mixing quality coefficient and is then independent of physical units. This allows the mixing qualities of a wide variety of products to be compared with one another. The minimum number of samples per batch should generally not be less than ten in order to ensure sufficient statistical significance of the homogeneity test.
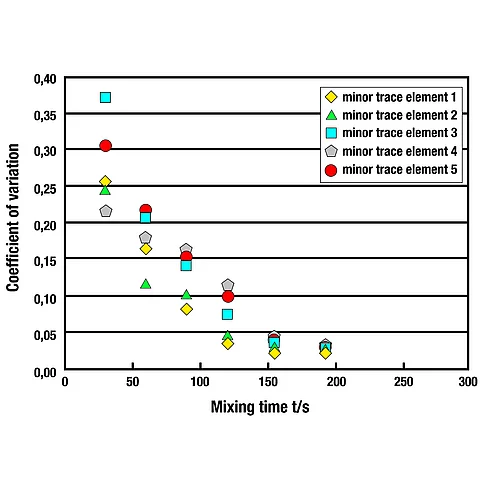
Practical example (amixon®)
In practice, tests are carried out with amixon® mixers using component ratios of up to 1:100,000, amongst other things. The particle size of the minor component typically lies in the range of approximately 10 to 50 micrometres, with mixing batches of around 3 cubic metres and sample quantities of only about 15 grams. In these tests, the quantity of the minor component is determined analytically; the coefficients of variation for mixing quality obtained are in the range of approximately 1.5 to 3.5 per cent.
These analytical data demonstrate that very high mixing qualities can be achieved reproducibly with precision mixers.

