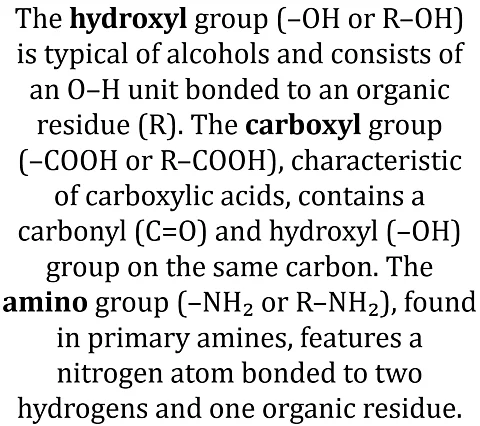
hydrophilic
Hydrophilic means "water-loving". The term describes a substance's affinity for water. Hydrophilic substances are easily wetted by water. They often dissolve in water or swell when immersed in it.
Hydrophilic molecules usually have polar or charged groups. Typical examples are hydroxyl, carboxyl or amino groups. These groups can form hydrogen bonds with water molecules. They can also enter into ionic interactions.
In formulation technology, this is referred to as hydrophilic systems or matrices. Such systems absorb water and swell. They can form gels and bind water over a longer period of time. This is important for thickening, stabilising and the controlled release of active ingredients.
Structural formulas of typical hydrophilic groups:
- Hydroxyl group: –OH or R–OH (e.g. in alcohols).
- Carboxyl group: –COOH (functional group of carboxylic acids).
- Amino group: –NH₂ or R–NH₂ (in primary amines).
These groups make many organic molecules hydrophilic because they have strong polar bonds and can form hydrogen bonds with water.

