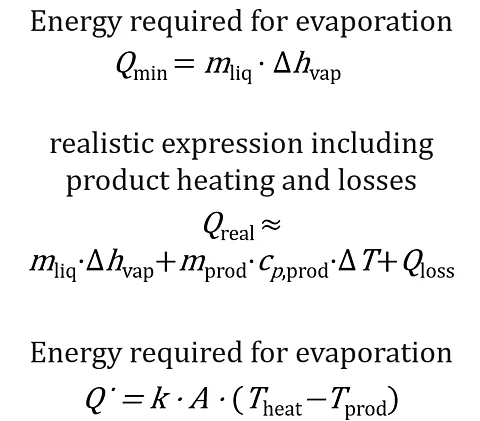
Vacuum contact drying
acuum contact drying is a drying process below atmospheric pressure. The product is in direct contact with heated wall surfaces or mixing tools. Due to the reduced pressure, the boiling point of the liquid decreases. This allows efficient drying at low temperatures.
Heat transport occurs predominantly through conduction and contact heat transfer. Convection plays only a minor role. The specific heat transfer area of the apparatus is therefore of great importance. A large contact area increases the drying rate and energy efficiency.
The heat requirement for evaporation is described by the following equation:
Qdot = mdot * Delta_h_vap
- Q_(dot) is the supplied heat power (W)
- m_(dot) is the mass flow rate of the evaporating water or solvent (kg/s).
- h_(vap) is the enthalpy of vaporization of the liquid (J/kg).
The drying rate results from the energy balance:
m_(dot,evap) = Q_(dot) / Δh_(vap)
- m_(dot,evap) is the evaporation rate of the liquid (kg/s)
- Qdot is the supplied heat power (W)
- Delta_h_vap is the enthalpy of vaporization (J/kg).
The heat flow across the heated wall can be described by the following equation:
Qdot = U * A * ΔT
- U is the overall heat transfer coefficient (W/(m²·K))
- A is the heat transfer area (m²)
- Delta_T is the temperature difference between the heating medium and the product (K)
Vacuum contact drying is used for powders, granulates, pastes, and suspensions. It is an important process in the chemical, food, pharmaceutical, and battery industries. The process is particularly suitable for temperature-sensitive, oxidation-prone products. Solvents can be recovered with high purity so that they do not enter the environment.

