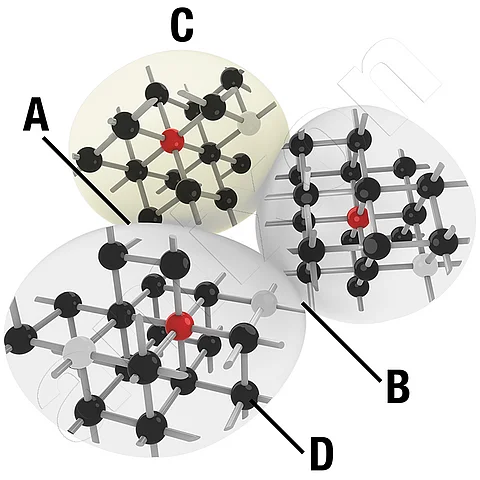
Impurity diffusion
Foreign diffusion refers to the diffusion of foreign atoms in a solid crystal lattice, that is, the migration of atoms that do not belong to the base lattice of the material. It occurs, for example, when an alloying element or dopant enters a metallic or ceramic base material and distributes within it. As a rule, a concentration gradient of the foreign atoms between two regions of the material is the driving force.
In contrast to self-diffusion, in which atoms of the same kind migrate within their own lattice, foreign diffusion describes the movement of atoms of a second substance in the base lattice. Typical mechanisms are site exchange via vacancies in the lattice (vacancy mechanism) or movement via interstitial sites (interstitial mechanism). The diffusion rate is determined by the temperature, the lattice structure, the defect density, and the concentration profile of the foreign atoms.
Technically, foreign diffusion is important in processes such as the heat treatment of alloys, the carburizing or nitriding of steels, the doping of semiconductors, or the formation of intermetallic phases at layer boundaries. Through controlled foreign diffusion, hardness profiles, corrosion resistance, electrical conductivity, and other material properties can be specifically adjusted.

